Generalized myasthenia gravis (gMG) is a heterogeneous autoimmune disorder in which antibody-mediated disruption of neuromuscular transmission leads to fluctuating weakness and variable treatment response. Management is evolving from broad immunosuppression toward more individualized approaches informed by autoantibody subtype, thymic pathology, and age of onset. Distinct immunologic patterns have been associated with key subgroups, including Th17-driven early-onset disease, immune-related changes in late-onset disease, complement-independent MuSK-positive disease, and heterogeneous mechanisms in seronegative forms.
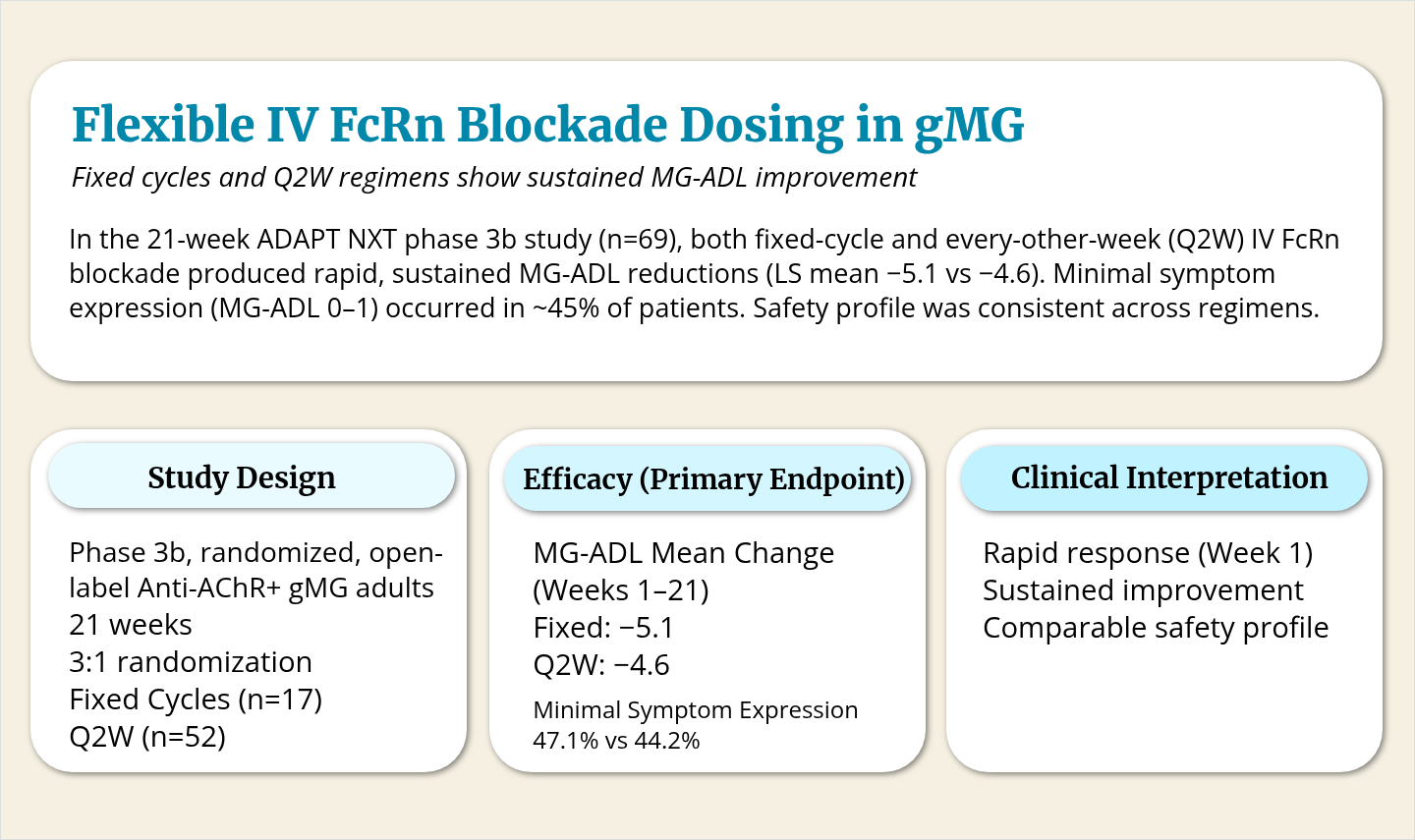
These insights have supported increased use of targeted biologics. Neonatal Fc receptor (FcRn) antagonists, which reduce pathogenic IgG levels, and complement inhibitors have been associated with improved outcomes in clinical studies of patients with refractory disease. Emerging cell-based and cytokine-directed therapies may further expand options for patients with highly refractory disease, although many remain investigational. Safety considerations, access, and monitoring requirements continue to influence treatment selection.
Highlights:
- gMG subtypes differ by antibody profile, thymic involvement, and immune pathway activation
- FcRn antagonists and complement inhibitors have been associated with improved outcomes in refractory disease
- MuSK-positive gMG is driven by IgG4 antibodies and often responds to B-cell–directed therapy
- CD19-, BAFF/APRIL-, BTK-, and cytokine-targeted therapies are under investigation
- CAR-T, CAAR-T, and stem cell approaches aim to achieve durable immune modulation
What Sets This Study Apart:
This review links immunopathology to treatment selection, highlighting how biologic and emerging therapies may be aligned with specific gMG subtypes rather than applied uniformly.
Limitations:
- Long-term comparative data across biologics remain limited
- Several advanced therapies are investigational or early phase
- Treatment access and biomarker standardization vary across settings
- Not all refractory patients respond despite targeted approaches
How are you incorporating antibody subtype and underlying immune mechanisms into treatment selection for gMG? What challenges do you face in applying targeted or emerging therapies in practice?